Freshwater Fingerprints: DataStream’s New guide for Reporting Isotope Data

Water is complex. In order to protect water, we need to answer a lot of questions. Where does water come from? How vulnerable is our water to pollution and climate change? What pollutants are in water, and where did they come from?
To answer these questions, scientists tap into a special feature that water has: its fingerprints. Water from different places has fingerprints, unique markers that can identify where the water comes from and how it moves through the environment.
One way to read these fingerprints is with isotopes. Isotope data is a powerful tool for assessing the quality and sustainability of water.
What is an isotope?
Every element on the periodic table has at least one isotope. Isotopes are forms of an element with the same number of protons and electrons but a different number of neutrons. Isotopes share the same chemical properties, but the difference in the number of neutrons makes each isotope have a different mass.
For example, an individual water molecule (H2O) is made of two hydrogen (H) atoms and one oxygen (O) atom. However, two water molecules are not always the same. Most water molecules have atoms made up of light isotopes and some have heavier isotopes.
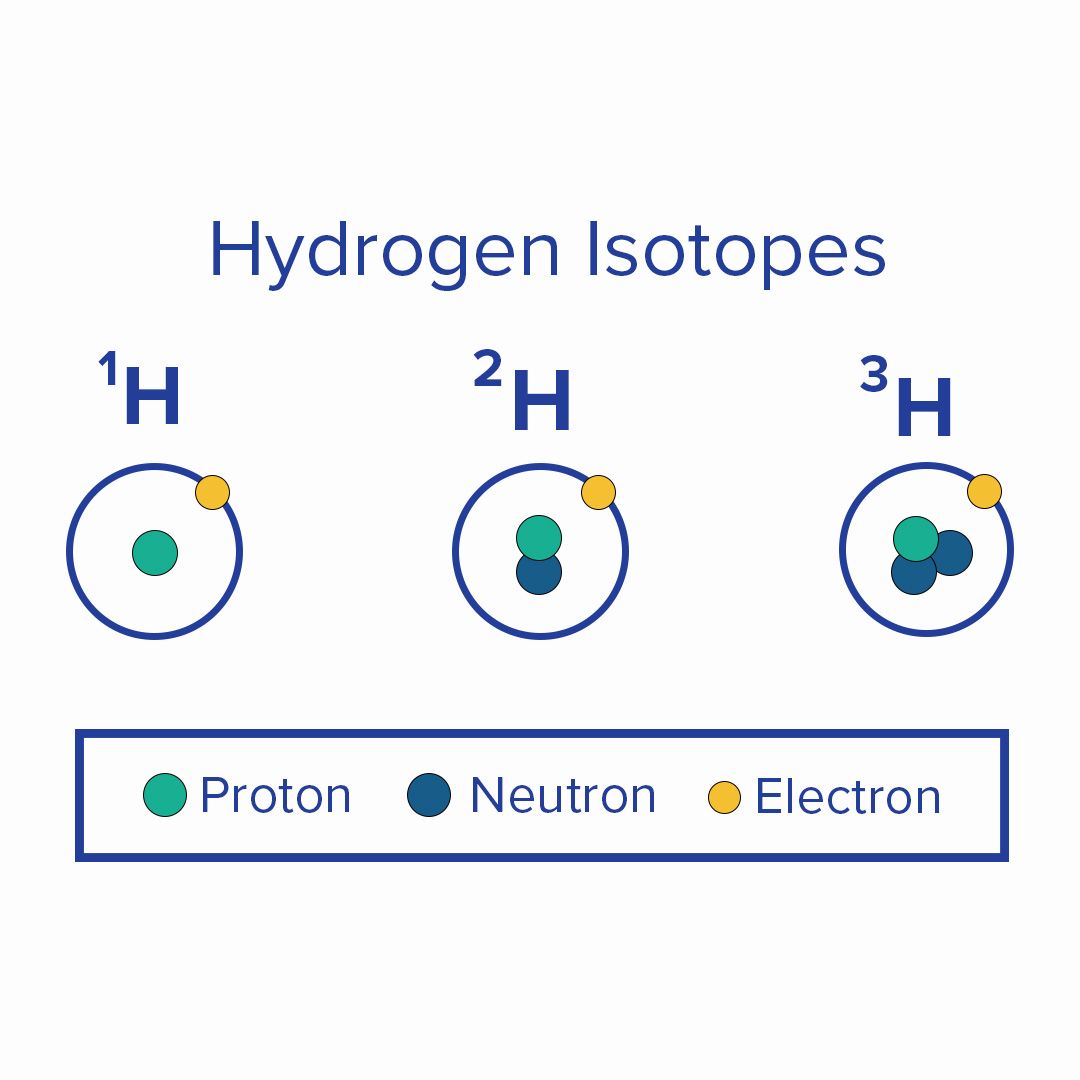
Isotopes are atoms with the same number of protons but different numbers of neutrons. Isotopes share almost the same chemical properties but differ in mass, and thus differ in physical properties. Hydrogen isotopes are called protium (1H), deuterium (2H), and tritium (3H).
Why measure isotopes?
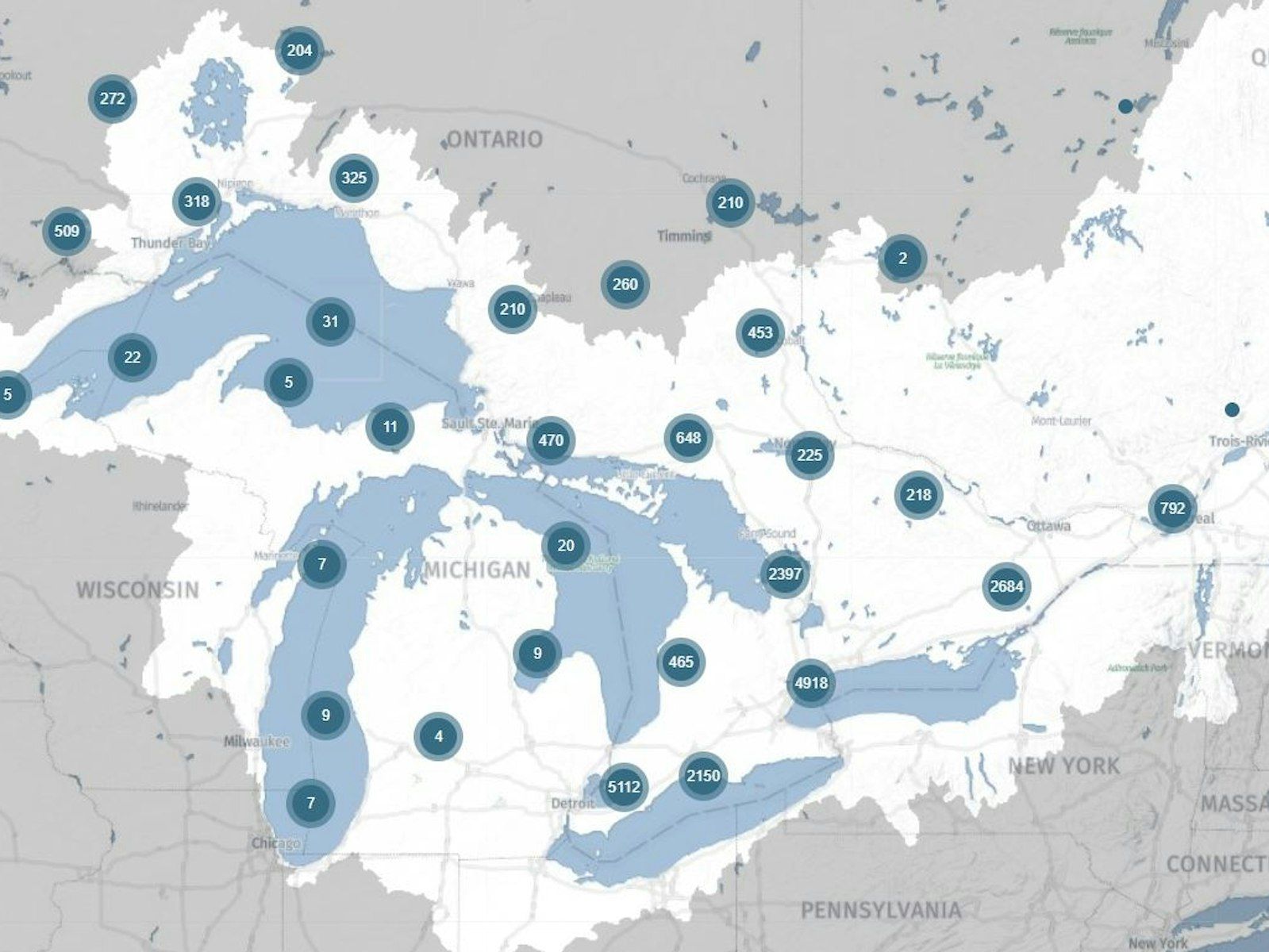
Measuring isotopes can tell us a lot about precipitation and where water comes from, which helps us properly evaluate water bodies. Isotope data can be used to differentiate surface water, groundwater, and glacial waters.
But isotope data can be used for so much more than tracing the origins of water. About 70% of food production worldwide depends on groundwater and it is the most abundant source of freshwater available to humans. Isotope data is the most direct and powerful tool to estimate the age and vulnerability of groundwater.
Different isotopes can be used to tell how old groundwater is; water with a detectable amount of an isotope called tritium may be about 60 years old, whereas carbon-14 is used to date water up to about 40 000 years old, and kryptom-81 is an isotope that can measure water up to a million years old.
“Old” groundwater has slow water flow, meaning that the aquifer that holds the groundwater will take a long time to replenish. Younger groundwater can be easily and quickly renewed by rainwater, but it is more vulnerable to pollution and climate change. Using isotopes to understand the age of water allows scientists and policy makers to manage groundwater more effectively.
Isotopes are also a powerful way to better understand water quality and pollution. Pollutants in water come from a variety of sources. By studying the isotopic composition of a pollutant, we can figure out where the pollutant originated.
For example, excess nitrogen is a common pollutant in waterways. Nitrogen pollution can contribute to lake eutrophication, overgrowth of plant life, and sometimes death of fish and animals from lack of oxygen in the water.
Nitrogen can enter waterways from sources such as fertilizers or human waste. Because nitrogen isotopes in agricultural fertilizer typically have different values from the nitrogen isotopes in human waste, scientists can measure the isotopes to know which source the pollutant originated from before entering a waterway.
What’s more, by measuring the nitrogen isotopes in aquatic insects, scientists can continue tracking the pollutant throughout the food web. This information can then be used to better understand and mitigate future pollution.
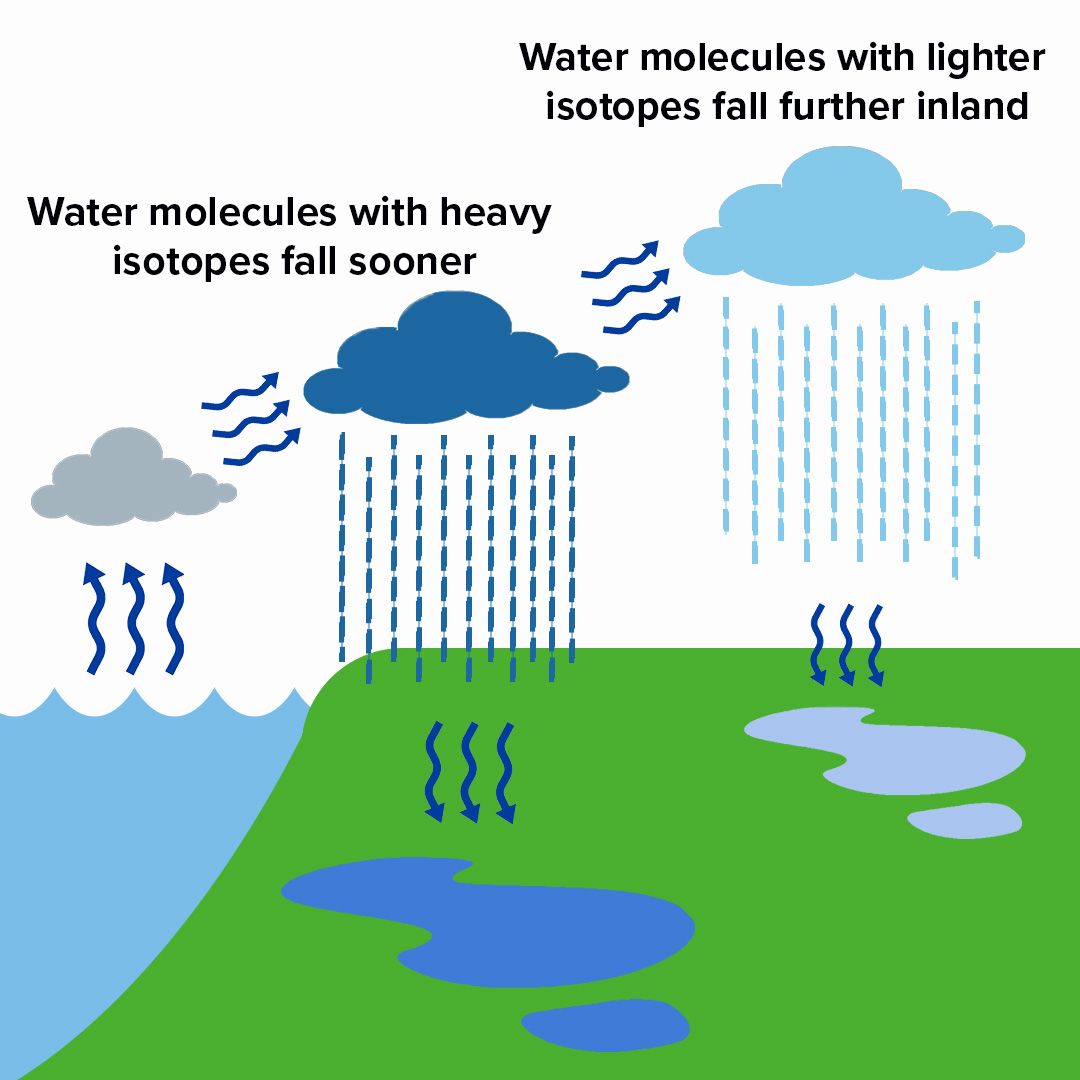
As water evaporates from the ocean, clouds form. As weather systems move inland, the water molecules with heavier isotopes condense first and fall as rain. Then, the lighter water molecules condense and fall. The further the clouds are from the coast, the less heavy isotopes they will contain. These isotopes can then be measured in groundwater and surface water. Graphic after IAEA Bulletin, 2019.
How are isotopes measured?
There are two types of isotopes: stable isotopes and radioisotopes (also known as unstable isotopes). Stable isotopes do not emit radiation, while radioisotopes undergo radioactive decay and do emit radiation.
Isotopes are measured by taking samples of water, soil, or biological materials, among other substances. The samples are then sent to a lab where they are prepared and measured using techniques such as mass spectrometry or laser spectroscopy. Isotopes of the following elements are commonly measured for water quality applications: oxygen (O), hydrogen (H), carbon (C), and nitrogen (N).
New DataStream Guide
Isotope data can be complex, and formatting isotope data can be a daunting task. That’s why we are so excited to release our new DataStream Guide: Best Practices for Reporting Isotope Data. The goal of the document is to support a consistent approach when handling isotope data.
Isotope data is a powerful tool for unlocking stories about environmental processes that impact water quality. Uploading isotope data to DataStream helps place these stories in the bigger picture. If you have isotope data that you'd like to publish on DataStream, please contact us, we'd love to work with you!