Freshwater generally has alkalinity in the range of 20-200 mg/L. However, alkalinity varies with differences in the geology (type of rocks) in an area.
What is it?
Alkalinity measures water’s ability to maintain a stable pH level. This is also called buffering capacity. The higher the alkalinity of a water body, the better it can protect itself against the addition of strong acids or bases, like chemical spills or contaminated surface runoff.
Total alkalinity is the sum of all components that act as a pH buffer in water. In natural freshwater systems that is mainly carbonates (CO32-), bicarbonates (HCO3-), and hydroxides (OH-). The measurement of only carbonates and hydroxides in the water is referred to as phenolphthalein alkalinity.
Alkalinity in natural river, lake and groundwater systems is mainly influenced by dissolved minerals like calcium carbonate (CaCO3) entering the water from soil and certain types of rocks. Whereas limestone contains calcium carbonate and will therefore contribute to higher alkalinity in water, rocks like granite do not.
Alkalinity can also be influenced by carbon dioxide from the atmosphere reacting with water. Human-driven sources that contribute to higher alkalinity include fertilizers that often contain broken down limestone rock.
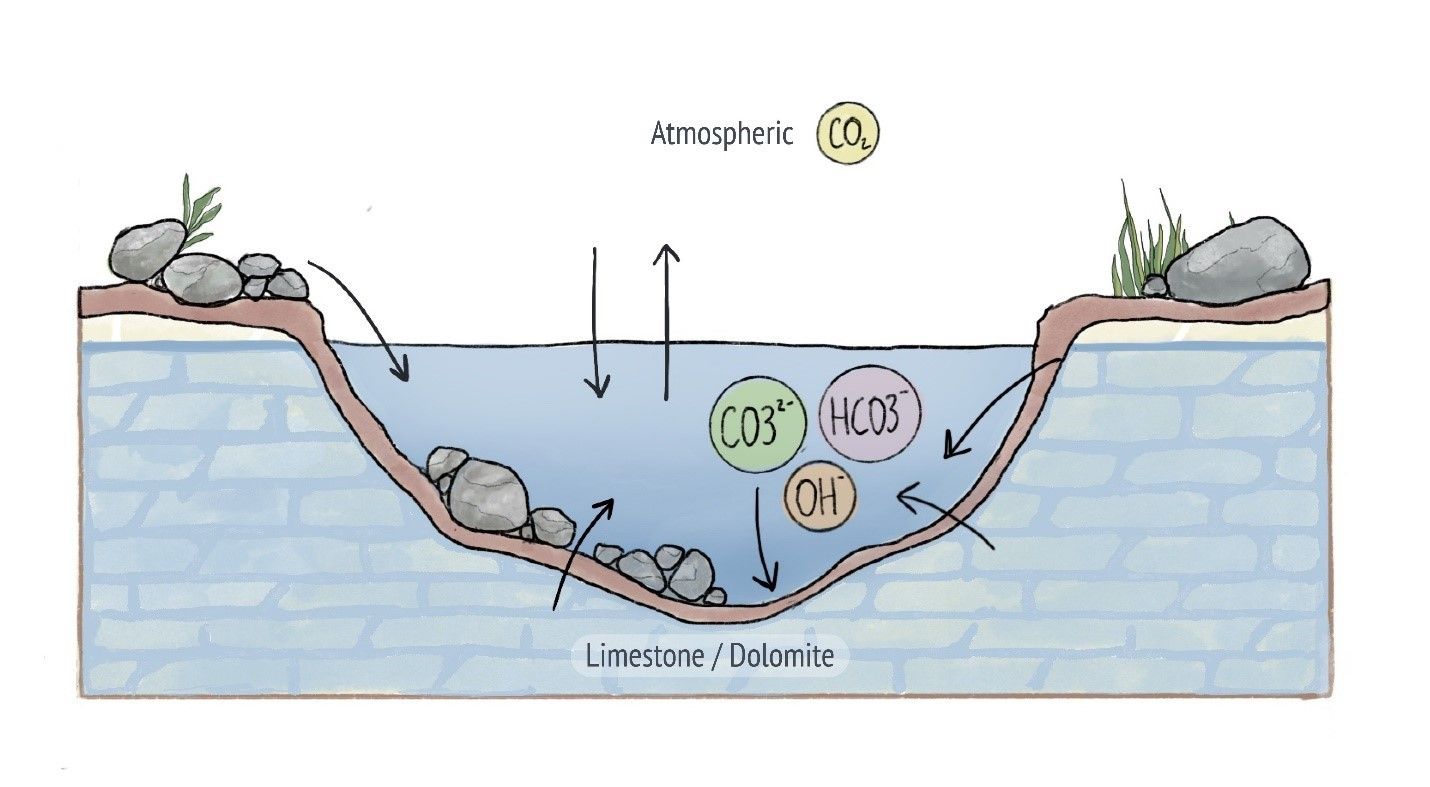
Alkalinity in water is influenced by minerals entering the water from soil and rock, as well as atmospheric carbon dioxide.
Why does it matter?
Fish and other organisms that live in water thrive in stable conditions. Water systems with high alkalinity tend to have more stable pH levels, thanks to alkalinity’s buffering effect. When a water body has low alkalinity and a sudden change occurs, such as a chemical spill or large addition of wastewater, it can lead to a rapid change in pH that is not healthy for fish and other organisms.
How is it measured?
Alkalinity is typically measured in a water sample sent to a laboratory, and is usually expressed as a calcium carbonate concentration.
A common method of measuring alkalinity is called alkalinity titration, which involves checking the pH of a water sample while small amounts of acid are added. The pH of the initial water sample is recorded, then each time a small amount of acid is added the pH value of the sample is recorded again. This is repeated until the carbonates in the water can no longer keep the pH stable and there is a sudden drop in pH. The total amount of acid added that results in a drop in pH is used to calculate the concentration of carbonate, and therefore alkalinity, of the water sample.
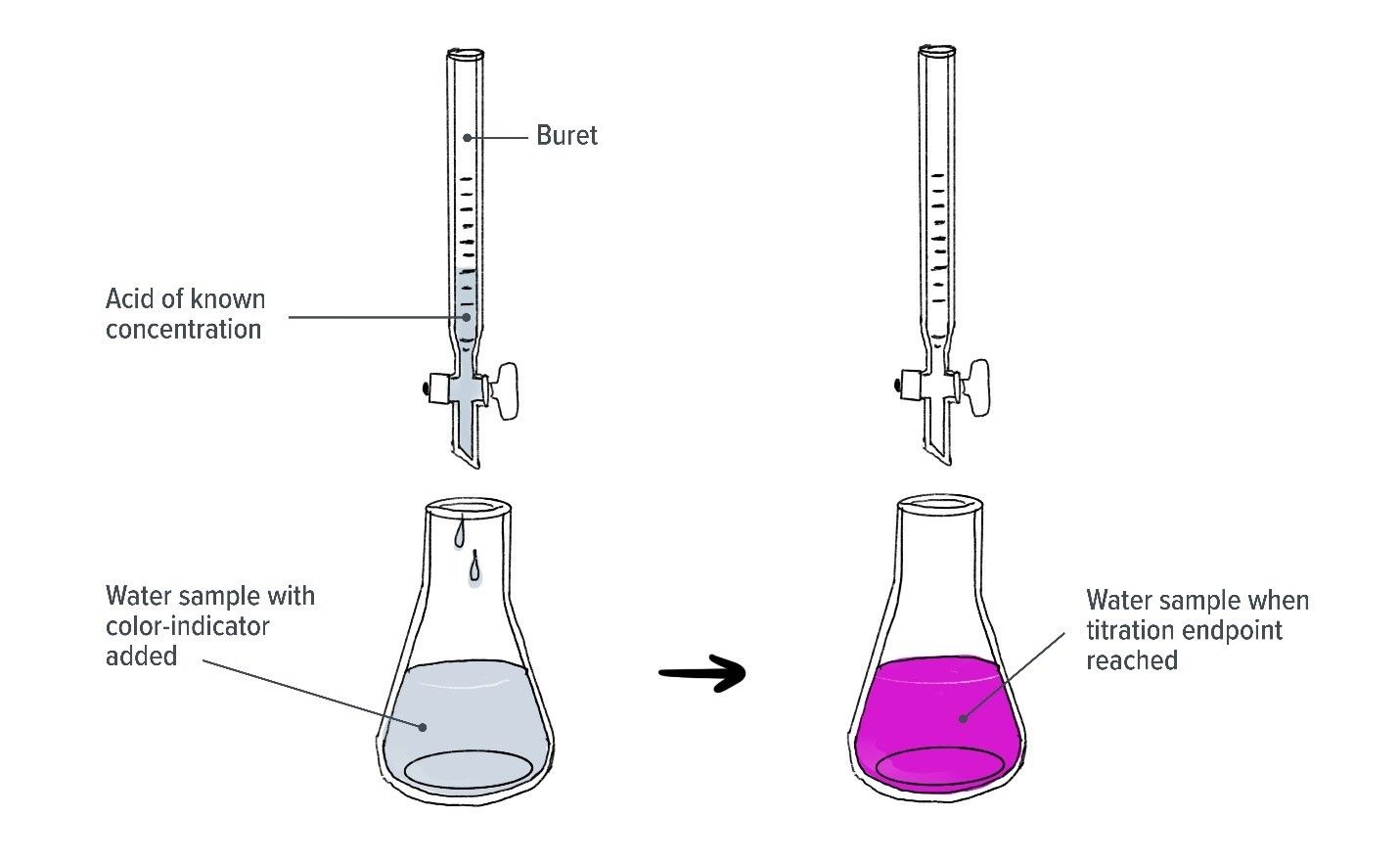
Alkalinity titration is a common method used to measure alkalinity. Small amounts of acid are added to a water sample until there is a sudden drop in pH. Some methods add an indicator substance to the water sample so that a colour change occurs when this happens.
Alkalinity influences: pH.