In rivers and lakes, total organic carbon concentrations are generally less than 10 mg/L, unless the water receives waste or is highly coloured due to natural organic material. Total organic carbon concentrations can be highly variable depending on the region.
What is it?
Carbon is a chemical element that is a common component of all life on Earth (organic carbon). Carbon is also abundant in the earth’s crust as carbonate minerals and rocks, and in the air (inorganic carbon).
Because of the unique properties of carbon, it can form more compounds than any other element, which means that it can be measured in many different ways in the environment.
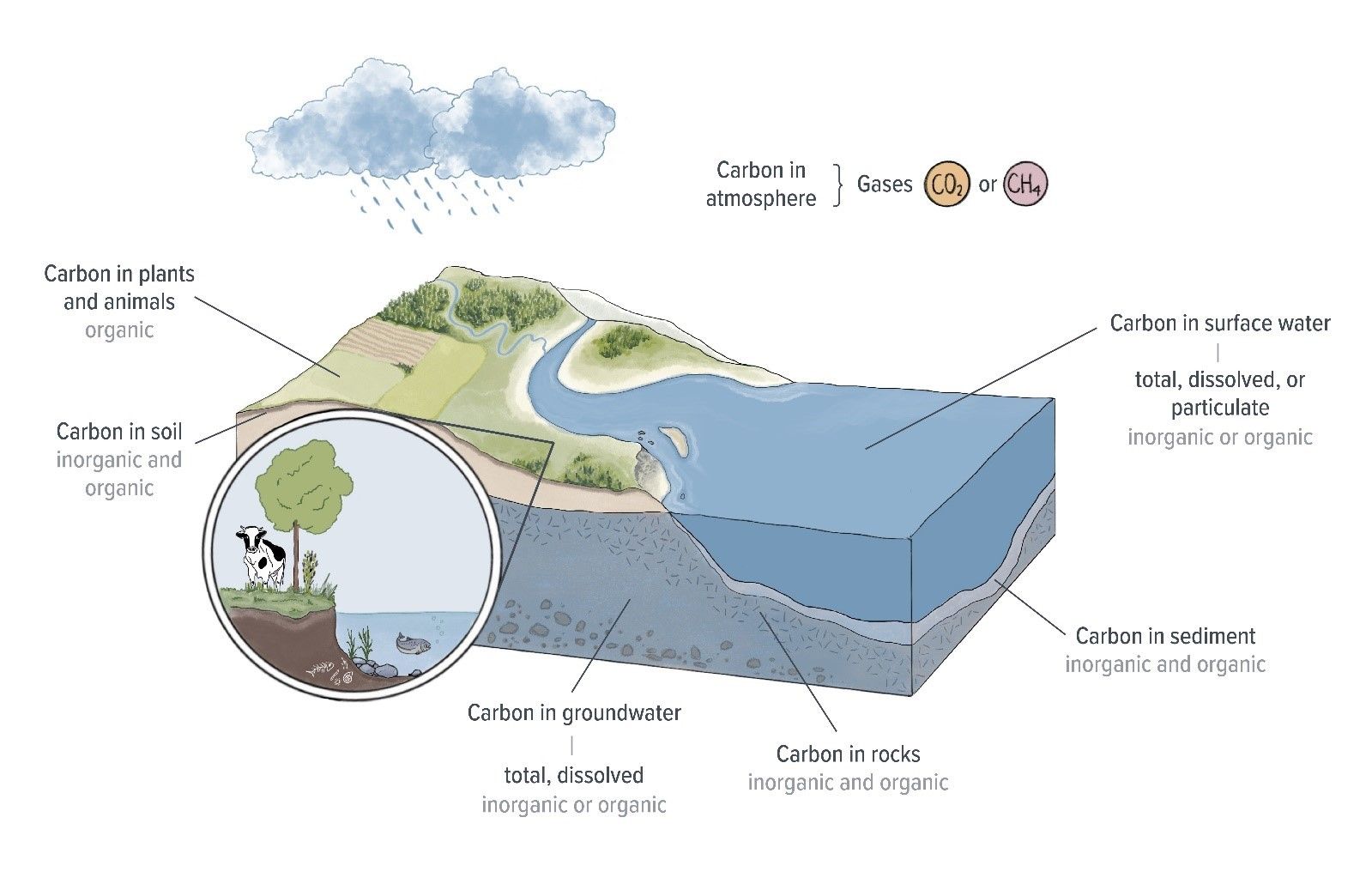
In lakes and rivers, carbon is present as a gas dissolved in water, as ions or particles in water, in sediments, and in all aquatic organisms from tiny plankton to large fish. Carbon also exists in the atmosphere and on land. Carbon coninually moves between all these different elements in what is called the carbon cycle.
Why does it matter?
Carbon influences the function of aquatic ecosystems in many different ways. Carbon serves as a source of energy and mass within aquatic food webs. Carbon also effects water colour, how susceptible a river or lake is to acidification, and the potential for accumulation of nutrients and certain contaminants in particles and sediments.
The abundance and forms of inorganic carbon in water is important because it provides a buffer to acidification, like acid rain or acid drainage. Inorganic carbon, like bicarbonate and carbonate, helps neutralize acid in water.

Water draining into a river or lake from the surrounding land can be a source of organic carbon that gives a brown tint to the water.
How is it measured?
Carbon is typically measured in water or sediment samples sent to a laboratory for analysis. Carbon can be measured as a gas, liquid, or solid (as particles).
Carbon is generally reported as being either organic (i.e., containing carbon-hydrogen bonds, or hydrocarbon) or inorganic (carbon dioxide, carbonic acid, bicarbonate, and carbonate). Carbon isotopes can also be measured in water and sediments, and are often used to determine the sources of carbon to lakes and rivers.
Carbon influences: water colour, water temperature, alkalinity, pH.
Carbon is influenced by: pH, chlorophyll.