National guidelines recommend less than 0.026 µg/L of mercury in water for the protection of aquatic life. The guideline for methylmercury is even lower, at 0.004 µg/L.
What is it?
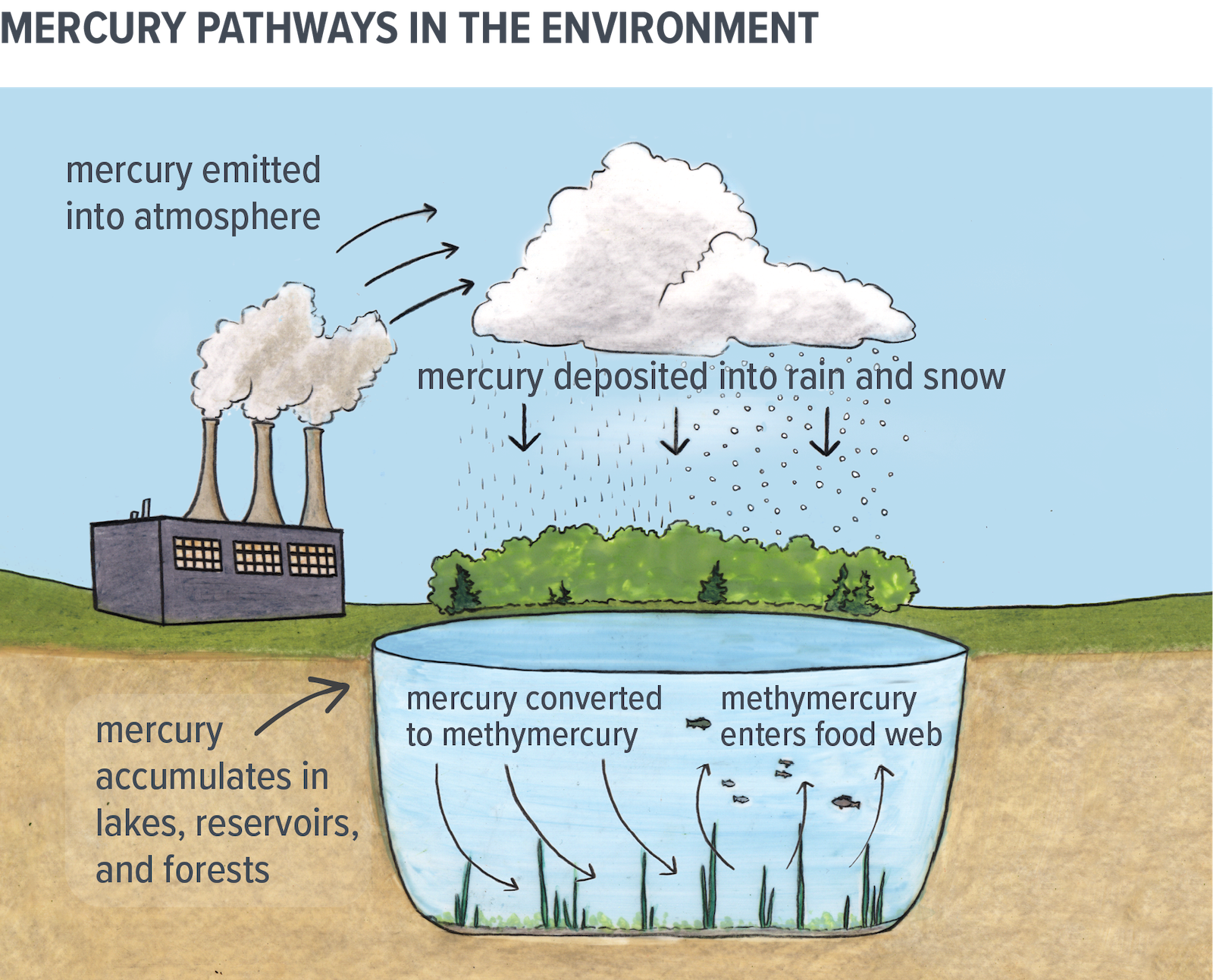
Mercury is a metal that is toxic and extremely persistent in the environment. Mercury is released naturally through weathering of rocks, forest fires, and volcanic activity. But human activity has contributed significantly to the amount of mercury in the environment. Coal-burning power plants, waste incineration, metal mining, and smelting all release mercury into the atmosphere.
In vapour form, mercury can travel long distances through the air before it is deposited on land and in water. Mercury in the atmosphere exists mostly as elemental mercury, one of its less-toxic forms. However, it can be converted to other forms once deposited onto the landscape.
Why does it matter?
Bacteria convert mercury to methylmercury, a more bioavailable and highly toxic form of mercury. This often happens in waters where minimal oxygen is present such as at the bottom of lakes and in wetlands, or where soils and other organic material become submerged (e.g. at newly created hydroelectric dam reservoirs).
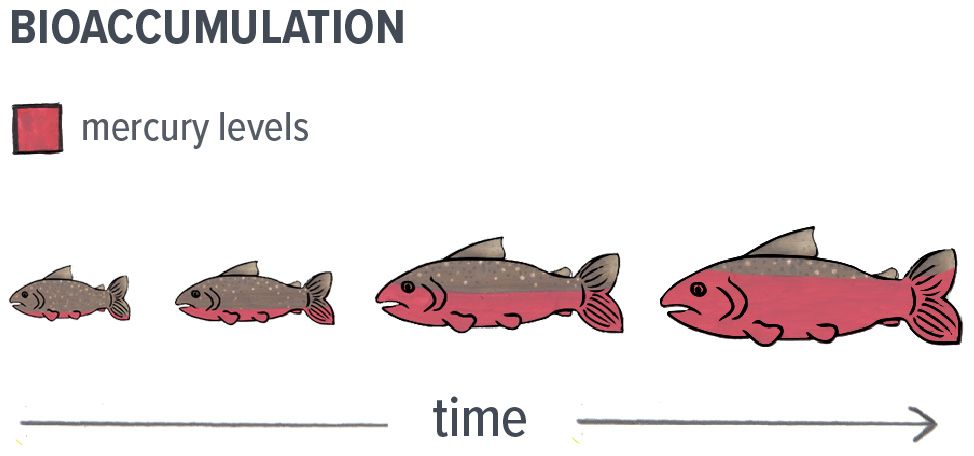
Once methylmercury is present in water bodies, it is easily absorbed by small aquatic organisms and fish. Since methylmercury does not break down in these organisms, it accumulates in their tissues over time and this is referred to as bioaccumulation.
It is important to measure mercury concentrations in water bodies, fish and animal tissues in order to understand the potential health risks to local animals and people who depend on fish (especially large predatory fish) for a significant part of their diet.
Methylmercury concentrations also progressively increase up the food chain in a process called biomagnification. This happens when large predatory fish eat smaller fish that contain methylmercury, and then birds and mammals (including humans) in turn eat these larger fish. The toxic effects of mercury primarily target the nervous systems (brain and nerves) of people and animals.
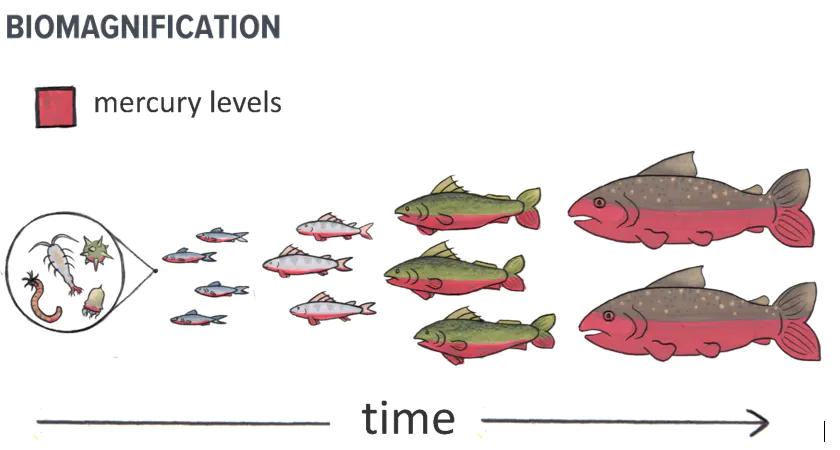
Larger predatory fish consume many smaller fish, accumulating mercury in their tissues. The older and larger the fish, the greater the potential for higher mercury concentrations in their bodies.
How is it measured?
Mercury and methylmercury samples are collected in the field in glass or specially coated plastic bottles, which are then sent to a lab for analysis. Specific procedures are used when collecting the sample to prevent contamination, which can easily occur.
A field-blank sample is also often taken, where a bottle filled with clean distilled water will go through all the steps as the others but will not be used to collect an actual sample. Analysis of this blank sample helps the lab determine if mercury contamination was present apart from the water samples taken. Mercury and methylmercury concentrations are often reported in very small units, such as nanograms per litre (ng/L).