Naturally occurring nitrate (NO3) concentrations in lakes and streams are typically less than 4 mg/L, and the concentration of nitrite (NO2) is generally much lower. The concentration of ammonia (NH3) in natural waters is generally less than 0.1 mg/L.
What is it?
Nitrogen, like phosphorus, is an important nutrient for plant growth. In rivers and lakes, nitrogen can be dissolved in the water, attached to particles floating in the water and found in the bodies of all living organisms.
Forms of nitrogen in freshwater systems include inorganic nitrogen: nitrate (NO3), nitrite (NO2), ammonia (NH3) and ammonium (NH4). These inorganic forms of nitrogen are the most bioavailable, meaning they are most easily used and taken up by organisms that live in the water. Other forms of nitrogen include organic nitrogen. Kjeldahl nitrogen includes ammonia, ammonium and organic nitrogen together.
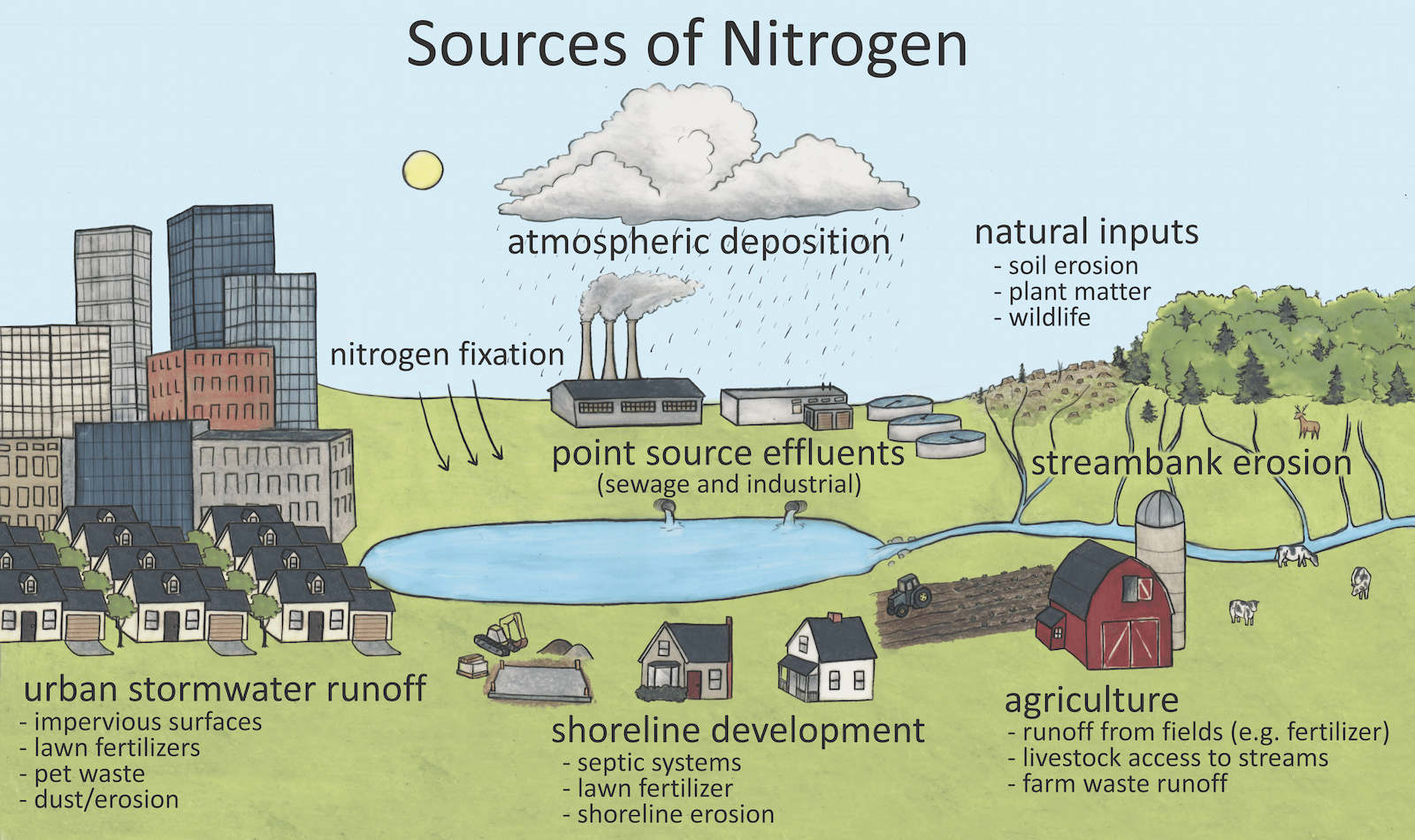
Why does it matter?
High concentrations of certain forms of nitrogen – including nitrate, and especially nitrite and ammonia – can be toxic to the organisms that live in the water. The toxicity of ammonia varies with pH and water temperature.
Nitrogen may also contribute to excess algae growth. However, research has found that in most cases reducing nitrogen inputs to a lake has little impact on algae blooms when phosphorus inputs continue. In estuaries, the role of nitrogen as a plant nutrient has been found to be more important, while in northern lakes and in some river systems, both phosphorus and nitrogen can act together as important plant nutrients.
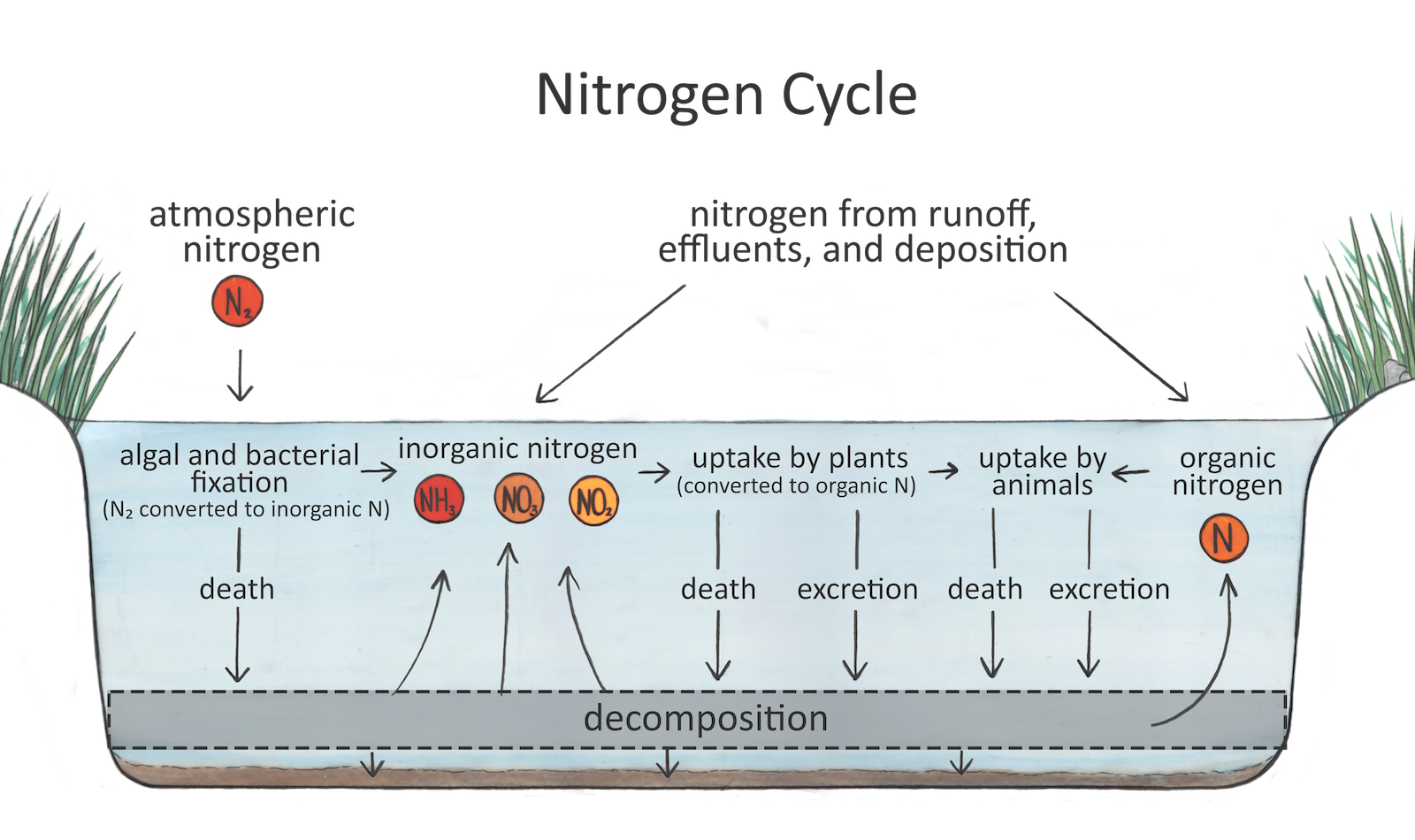
Nitrogen is converted into different forms as it cycles through aquatic ecosystems.
How is it measured?
Nitrogen is measured from a water sample sent to the lab. The sample may or may not be filtered before analysis. It is important to know what form of nitrogen is being measured, because the more bioavailable inorganic forms (nitrate, nitrite and ammonia/ammonium) behave differently and have different impacts on water quality compared to organic forms.
Related to nitrogen: water colour, total suspended solids (TSS).
Nitrogen influences: chlorophyll.